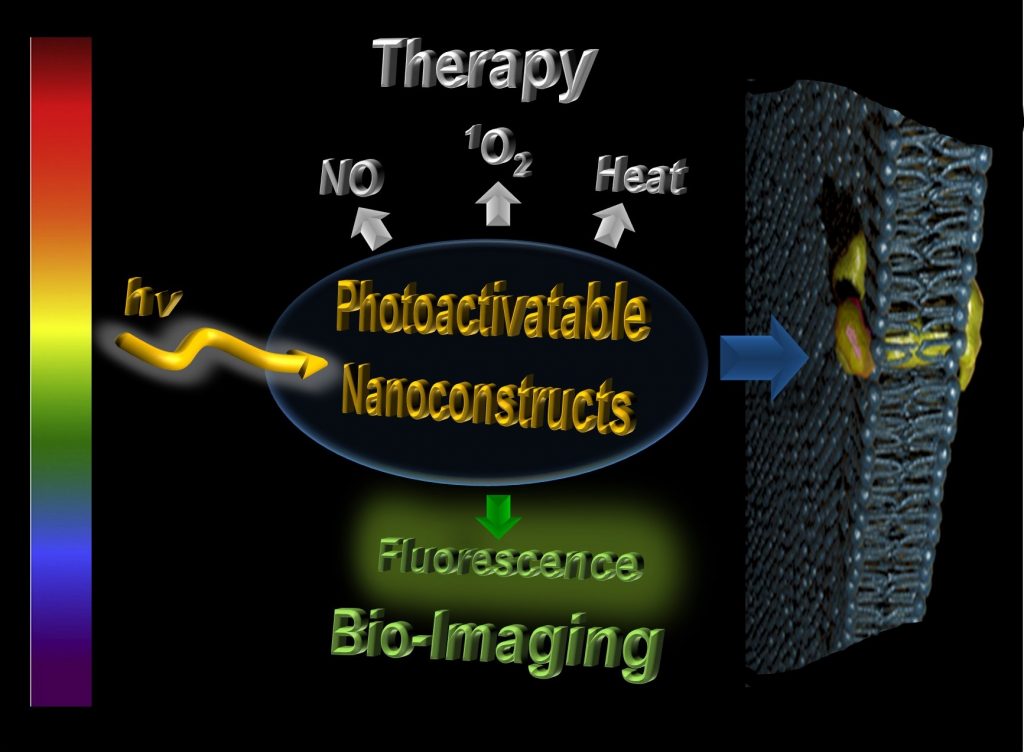
Why Combine Light and Nanomedicine?
The achievement of nanoconstructs able to release therapeutic species in a controlled fashion and to be tracked in a biological environment is a major challenge in the burgeoning field of nanomedicine. Light is a powerful tool to fulfil these needs in a non-invasive way. By virtue of its easy manipulation in terms of energy, intensity, location and duration, light mimics an “optical syringe” that can activate fluorescence for imaging and stimulate a “burst” of therapeutics with superb spatiotemporal control, by using tailored photoresponsive systems. Besides their key role in the activation of therapeutic and imaging functionalities, photons represent powerful and “green” reactants for the facile synthesis of noble metal-based nanomaterials, which are suited for a variety of bio-applications due to their characteristic plasmonic absorption.
Research Interests
Photoactivatable Nanoconstructs for Biomedical Applications
Our current research aims at the design and fabrication of “smart” nanoconstructs able to release Nitric Oxide (NO), Singlet Oxygen (1O2) and “Heat” under the exclusive control of light stimuli. All these species represent intriguing “unconventional” therapeutics for anticancer and antibacterial applications and play a key role in the NO-stimulated, photodynamic and photothermal therapy. In our laboratory, we have been working on the design and fabrication of a number of nanoconstructs, including molecular hybrids, polymeric and mesoporous silica nanoparticles, quantum dots, hydrogels, thin films and differently shaped gold and silver nanostructures, that can be activated, synthesized or both by using Visible/Near Infrared light as suitable trigger and that also exhibit fluorescence, which is useful for bio-imaging. Particular emphasis is given to systems able to photogenerate simultaneously more than one therapeutic agent in view of multimodal therapeutic treatments exploiting additive/synergistic effects. For some representative and recent examples click on the boxes below.
Selected Reviews:
- S. Sortino, Light-controlled nitric oxide delivering molecular assemblies, Chem. Soc. Rev., 2010, 39, 2903-2913.
- S. Sortino, Photoactivated nanomaterials for biomedical release applications, J. Mater. Chem. 2012, 22, 301-318.
- S. Sortino, F. M. Raymo et al. Photoresponsive polymer nanocarriers with a multifunctional cargo, Chem. Soc. Rev. 2014, 43, 4167-4178.
- A. Fraix and S. Sortino, Photoactivable platforms for nitric oxide delivery with fluorescence imaging, Chem. Asian J. 2015, 10, 1116-1125.
- F. Quaglia and S. Sortino, Polymer Nanoparticles for Photodynamic Therapy Combined with Nitric Oxide release and Chemotherapy, Appl. Photochem. 2016, 92, 397.
- A. Fraix, N. Marino and S. Sortino, Phototherapeutic Release of Nitric Oxide with Engineered Nanoconstructs, Topics in Curr. Chem. 2016, 370, 225-257.
- A. Fraix and S. Sortino, Combination of PDT photosensitizers with NO photodonors, Photochem. Photobiol. Sci. 2018, 17, 1709-1727.

A novel NO photodonor operates by the excitation with the highly biocompatible green light of the widely used chemotherapeutic doxorubicin as light-harvesting antenna without precluding its typical red emission and the DNA binding properties.
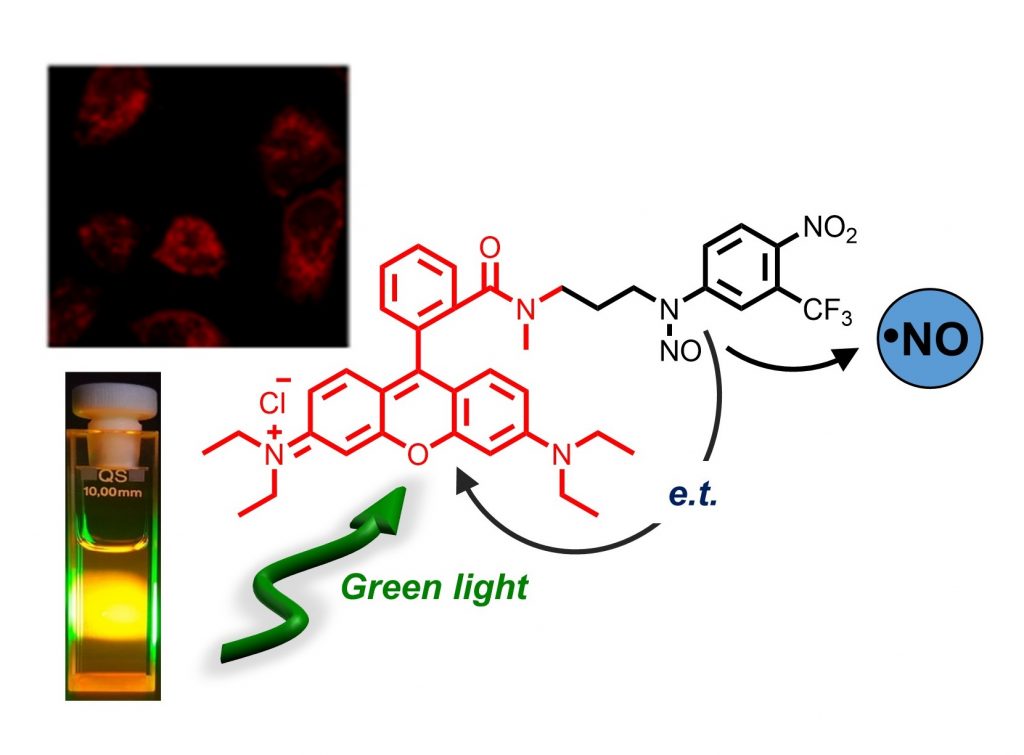
Green light excitation of the rhodamine unit triggers its red-orange fluorescence, useful to monitor the preferential mitochondrial accumulation, and encourages the NO release from the nitroso appendage for cytotoxic effects on cancer cells.
Selected Articles:
- A. Fraix, C. Parisi, M. Failla, K. Chevaev, F. Spyrakis, L. Lazzarato, R. Fruttero, A. Gasco and S. Sortino, NO release regulated by doxorubicin as green light-harvesting antenna, Chem. Commun. 2020, 56, 6332–6335.
- C. Parisi, M. Failla, A. Fraix, B. Rolando, E. Gianquinto, F. Spyrakis, E. Gazzano, C. Riganti, L. Lazzarato, R. Fruttero, A. Gasco and S. Sortino
Fluorescent nitric oxide photodonors based on BODIPY and rhodamine antennae, Chem. Eur. J. 2019, 25, 11080-11084. - M. Blangetti, A. Fraix, L. Lazzarato, E. Marini, B. Rolando, F. Sodano, R. Fruttero, A. Gasco and S. Sortino, A novel nonmetal-containing nitric oxide releaser activatable with single-photon green light, Chem. Eur. J. 2017, 23, 9026-9029.
- K. Chegaev, A. Fraix, E. Gazzano, G. E. F. Abd-Ellatef, M. Blangetti, B. Rolando, S. Conoci, C. Riganti, R. Fruttero, A. Gasco and S. Sortino, Light-Regulated NO Release as a Novel Strategy to Overcome Doxorubicin MultiDrug Resistance, ACS Med Chem. Lett. 2017, 8, 361−365.
- C. Parisi, M. Failla, A. Fraix, A. Rescifina, B. Rolando, L. Lazzarato, V. Cardile, A. C. E. Graziano, R. Fruttero, A. Gasco and S. Sortino, A molecular hybrid producing simultaneously singlet oxygen and nitric oxide by single photon excitation with green light, Bioorg. Chem., 2019, 85, 18-22.
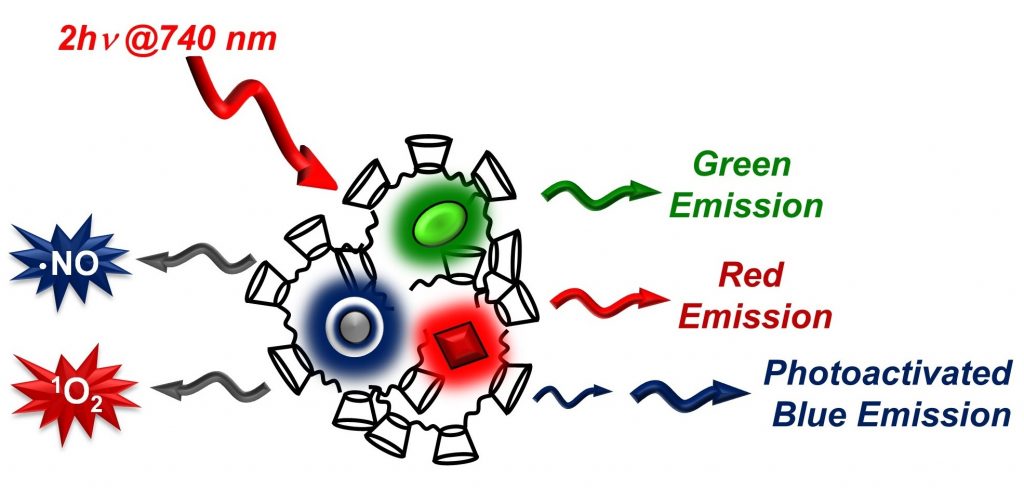
A multicomponent supramolecular nanoassembly based on a cyclodextrin-branched polymer can be activated by two-photon excitation with NIR light at 740 nm. This allows the simultaneous activation of five distinct functionalities, that is, the generation of green, red, and blue fluorescence for bio-imaging, and the production of 1O2 and NO for bimodal anticancer therapeutic effects.
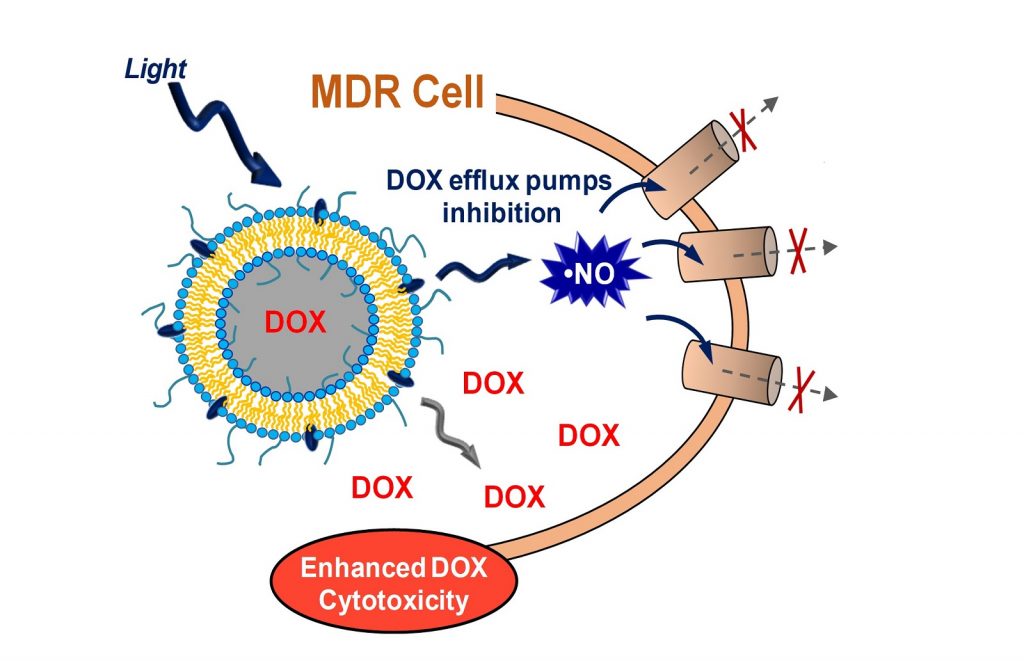
Tailored lipid-polymer hybrid nanoparticles (NPs) deliver nitric oxide under visible light stimuli, overcoming Doxorubicin (DOX) resistance through efflux pumps inhibition of DOX resistant cancer cells.
Selected Articles:
- A. Fraix, C. Conte, E. Gazzano, C. Riganti, F. Quaglia and S. Sortino, Overcoming Doxorubicin Resistance with Lipid-Polymer Hubrin Nanoparticles Photoreleasing Nitric Oxide, Mol. Pharm. 2020, in press
- A. Fraix, V. Kirejev, M. Malanga, E. Fenivesi, S. Beni, M. B. Ericson and S. Sortino, A three-color fluorescent supramolecular nanoassembly of phototherapautics activatable by two-photon excitation with near infrared light, Chem. Eur. J. 2019, 23, 7091-7095.
- F. Sodano, R. J. Cavanagh, A. K. Pearce, L. Lazzarato, B. Rolando, A. Fraix, T. F. Abelha, C. E. Vasey, C. Alexander, V. Taresco and S. Sortino, Enhancing Doxorubicin Anticancer Activity with a Novel Polymeric Platform, Biomater. Sci. 2020 in press.
- A. L. Tessaro, A. Fraix, M. Failla, V. Cardile, A. C. E. Graziano, B. M. Estevão, A. Rescifina and S. Sortino, Light-Controlled Simultaneous “On Demand” Release of Cytotoxic Combinations for Bimodal Killing of Cancer Cells, Chem. Eur. J. 2018, 24, 7664-7670.
- C. Conte, A. Fraix, H. Thomsen, F. Ungaro, V. Cardile, A.C.E. Graziano, M. B. Ericson, F. Quaglia and S. Sortino, Monitoring the Release of a NO Photodonor from Polymer Nanoparticles via Förster Resonance Energy Transfer and Two-Photon Fluorescence Imaging, J. Mater. Chem. B 2018, 6, 249-256.
Silica Nanoparticles
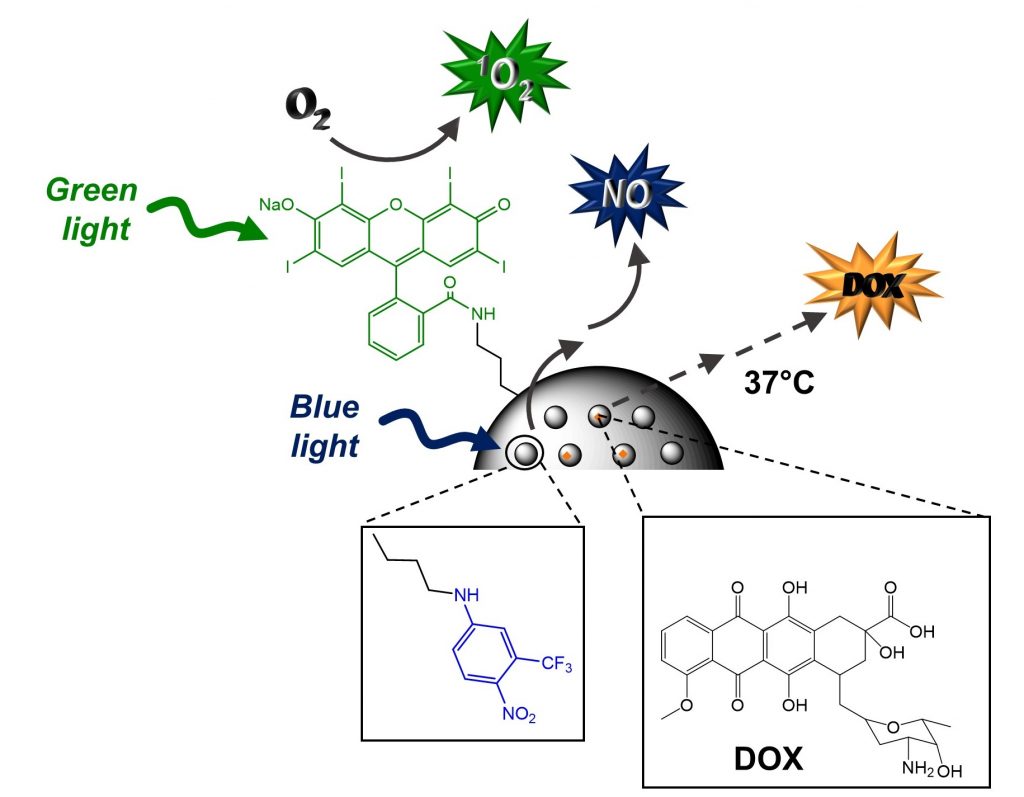
A “three bullet” nanoconstruct based on mesoporous silica nanoparticles for potential combined cancer photo-chemotherapy is able to generate 1O2 and NO under selective excitation with green and blue light, respectively and release the non-covalently entrapped anticancer DOX under physiological conditions.

Mesoporous silica nanoparticles release NO under light control and exploit the embedded carbon dots as fluorescent reporters for the NO liberated. These silica nanoparticles are also able to encapsulate the highly hydrophobic photosensitizer Temoporfin, preserving the fluorescent reporting function.
Selected Articles:
- A. L. Tessaro, A. Fraix, A. C. Pedrozo da Silva, E. Gazzano, C. Riganti and S. Sortino, “Three-Bullets” loaded mesoporous silica nanoparticles for combined photo/chemoterapy, Nanomaterials, 2019, 9(6). pii: E823
- D. Afonso, S. Valetti, A. Fraix, C. Bascetta, S. Petralia, S. Conoci, A. Feiler and S. Sortino, Multivalent mesoporous silica nanoparticles photo-delivering nitric oxide with carbon dots as fluorescent reporters, Nanoscale 2017, 9, 13404-13408.
- C. Zhou, D. Afonso, S. Valetti, A. Feiler, V. Cardile, A. C. E. Graziano, S. Conoci and S. Sortino, Targeted photodynamic therapy with a folate/sensitizer assembly produced from mesoporous silica, Chem. Eur. J. 2017, 23, 7672-7676.
- C. Fowley, A. P. McHale, B. McCaughan, A. Fraix, S. Sortino and J. F. Callan, Carbon quantum dots-NO photoreleaser nanohybrids for two-photon phototherapy of hypoxic tumors, Chem. Commun. 2015, 51, 81-84.
- V. Rizzi, J. Gubitosa, P. Fini, F. Fanelli, A. Fraix, S. Sortino, A. Agostiano, L. De Cola, A. Nacci and P. Cosma, A comprehensive investigation of amino grafted mesoporous silica nanoparticles supramolecular assemblies to host photoactive Chlorophyll a in aqueous solution, J. Photochem. Photobiol.: A Chem. 2019, 377, 149-158.
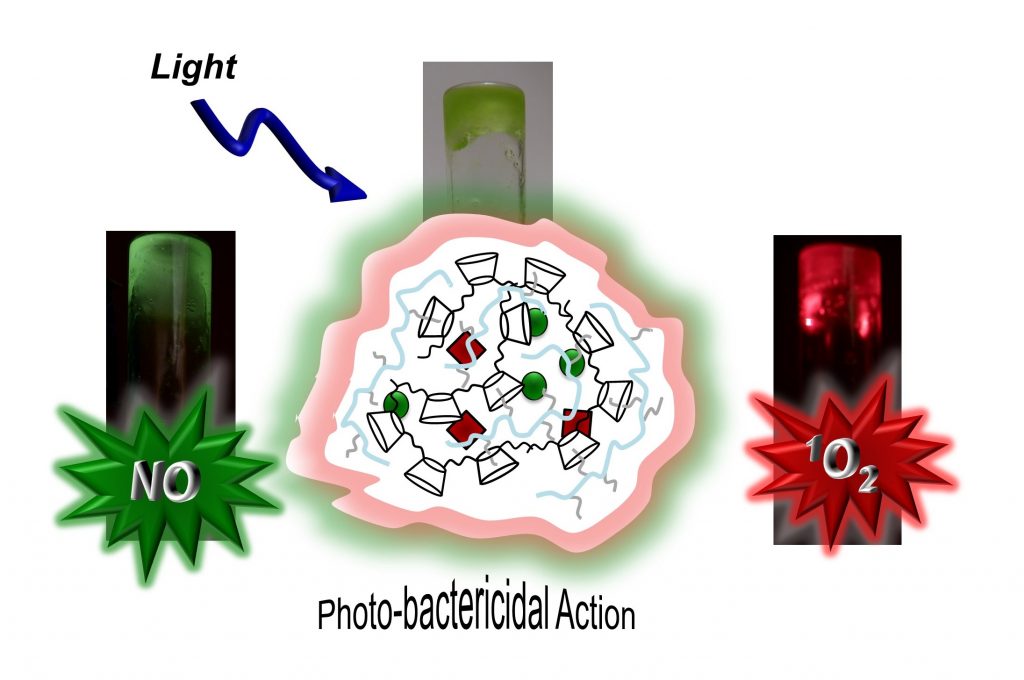
A multitasking photoresponsive supramolecular gel is easily achieved by the self-assembling of two polymeric components and two chromo-fluorogenic centers. Excitation with visible light results in the red and green fluorescence emission of the gel and in its capability to generate 1O2 and NO as bactericidal agents.
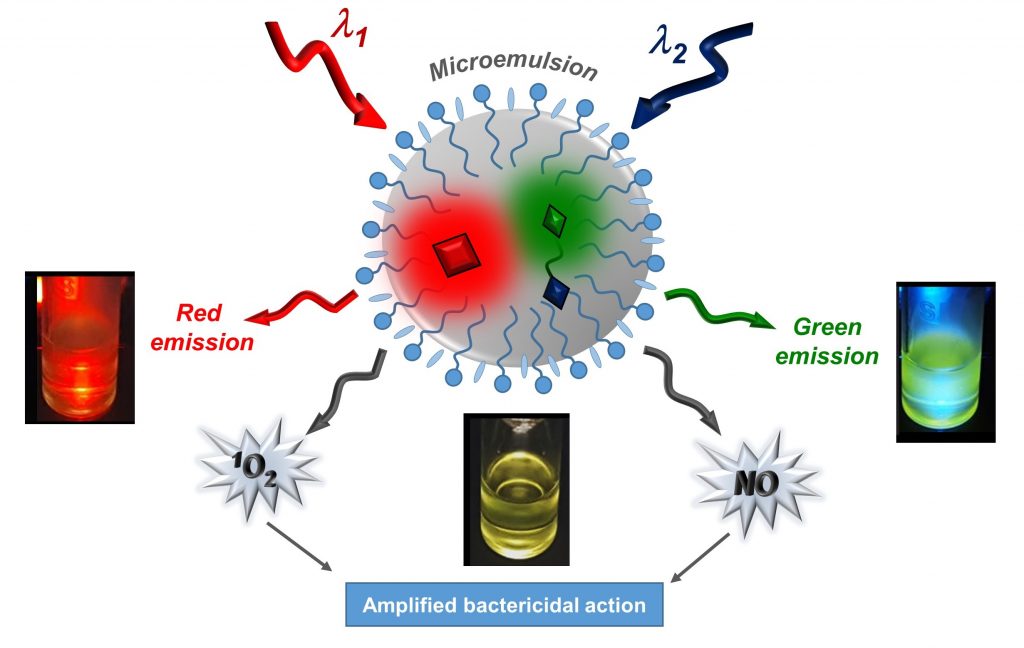
A microemulsion co-solubilizing a photosensitizer and a NO photodonor in the oily phase can be selectively excited with visible light stimuli of different wavelength resulting in the photogeneration of the cytotoxic 1O2 and NO, red and green fluorescence emission and amplified photobactericidal action.
Selected Articles:
- A. Fraix, O. Catanzano, I. Di Bari, C. Conte, M. Seggio, C. Parisi, A. Nostro, G. Ginestra, F. Quaglia and S. Sortino, Visible light-activatable multicargo microemulsions with bimodal photobactericidal action and dual colour fluorescence, J. Mater. Chem. B 2019, 7, 5257–5264
- A. Fraix, R. Gref and S. Sortino, A Multi-Photoresponsive Supramolecular Hydrogel with Dual-Colour Fluorescence and Dual-Modal Photodynamic Action, J. Mater. Chem. B. 2014, 2, 3443-3449.
- A. Fraix, N. Kandoth, R. Gref and S. Sortino, A multicomponent gel for nitric oxide photorelease with fluorescent reporting, Asian J. Org. Chem. 2015, 4, 256-261.
- N. Kandoth, J. Mosinger, R. Gref and S. Sortino, A NO photoreleasing supramolecular hydrogel with bactericidal action, J. Mater. Chem. B. 2013, 1, 3458-3463.

A NO photoreleaser with a fluorescent reporter has been integrated in the biocompatible PLGA resulting in a photoactivatable polymeric film. The NO release process can be activated by sunlight and can be followed in real time by the intense blue fluorescence the optical reporter. Once photogenerated, the NO radical promptly diffuses out of the polymeric matrix to reach biological target and induces excellent bactericidal activity.

The same principle shown left is extended to soft contact lens. The fluorescent reporting functionality, which activates concomitantly to the NO photorelease, allows the easy monitoring of the NO delivery in real time and confirms the working of the doped lens under daylight exposure. The NO photoreleasing lens are well-tolerated in both dark and light conditions by corneal cells while being able to induce good bacteria growth inhibition under visible light irradiation.
Selected Articles:
- N. Marino, M. Perez-Lloret, A. R. Blanco, A. Venuta, F. Quaglia and S. Sortino, Photo-antimicrobial polymeric films releasing nitric oxide with fluorescent reporting under visible lightl, J. Mater. Chem. B 2016, 4, 5138-5143.
- M. Seggio, A. Nostro, G. Ginestra, F. Quaglia and S. Sortino, Contact lens delivering nitric oxide under daylight for reduction of bacterial contamination, Int. J. Mol. Sci., 2019, 20, 3735-2743.
- J. Donansky, P. Henke, P. Kubat, A. Fraix, S. Sortino and J. Mosinger, Polystyrene nanofiber materials for visible light-driven dual antibacterial action via simultaneous photogeneration of NO and O2(1Dg), ACS Appl. Mater. Interf. Sci. 2015, 7(41), 22980-22989.
- G. Giancane, L. Valli and S. Sortino, Dual Funtion Multilayers for the Photodelivery of Nitric Oxide and Singlet Oxygen, Chem. Phys. Chem. 2009, 10, 3077-3082.
- E. Vittorino, G. Giancane, D. Manno, A. Serra, L. Valli and S. Sortino, Photofunctional multilayer films by assembling naked silver nanoparticles and a nitric oxide photodispenser at the air/water interface, J. Colloids Interf. Sci. 2012, 368, 191-196.
Noble Metal Nanostructures
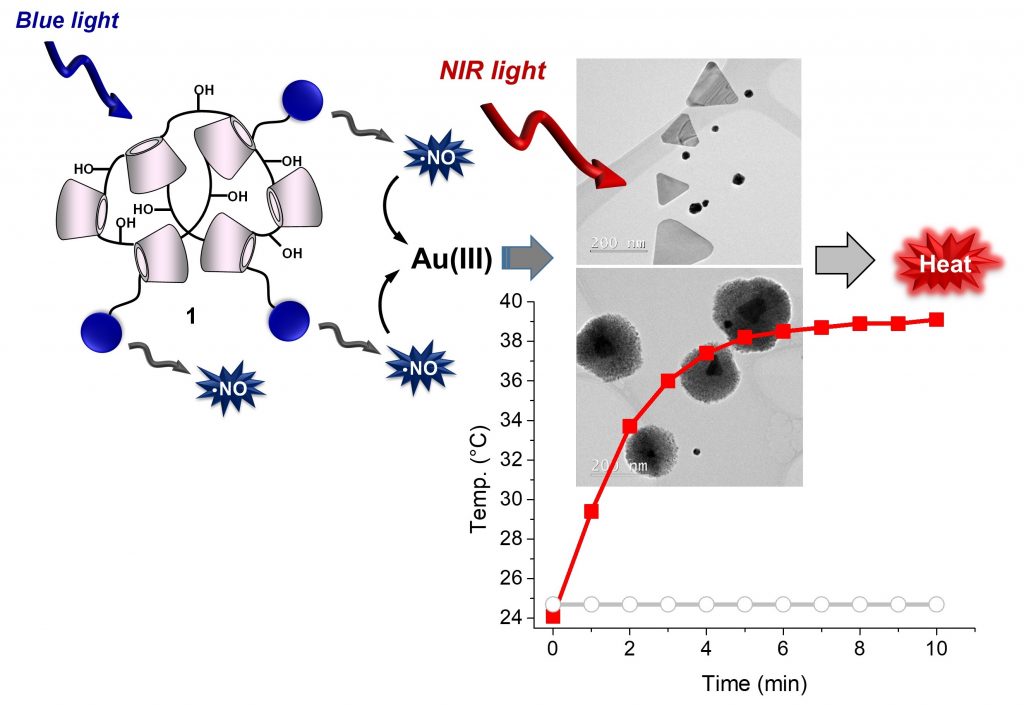
Water-dispersible Au nanotriangles and nanoflowers with NIR localized surface plasmon absorption are obtained by a simple “green” procedure based on the NO generated by visible light irradiation of a tailored cyclodextrin-branched polymer. Excitation of these nanostructures with 808 nm NIR light induces photothermal effects useful for cancer cell death and enzymatic reaction activation.
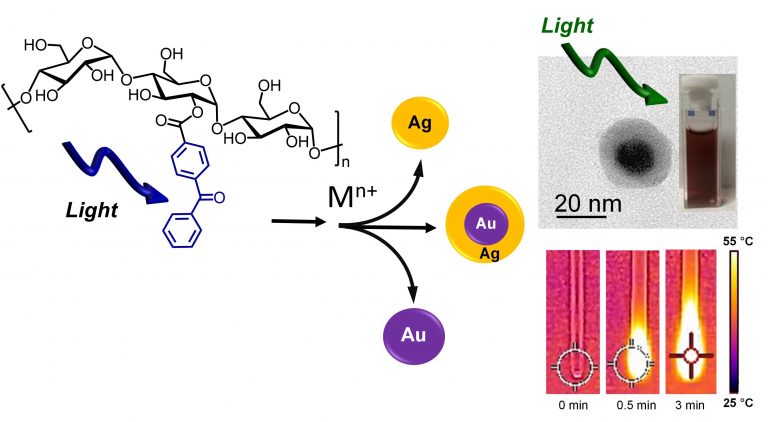
Water-dispersible Ag, Au and Au@Ag core-shell nanoparticles are rapidly and effectively obtained through the very fast generation of the highly reducing ketyl radical upon light irradiation of a biocompatible dextran functionalized with benzophenone. The resulting nanoparticles shows excellent antibacterial action and remarkable photothermal activity.
Selected Articles:
- G. Nocito, S. Petralia, M. Malanga, S. Beni, G. Calabrese, R. Parenti and S. Sortino, Biofriendly Route to Near-Infrared-Active Gold Nanotriangles and Nanoflowers through Nitric Oxide Photorelease for Photothermal Applications, ACS Appl. Nano Mater. 2019, 2, 7916-7923.
- M. Perez-Lloret, A. Fraix, S. Petralia, S. Conoci, V. Tafani, G. Cutrone, A. Vargas-Berenguel, R. Gref and S. Sortino, One-Step Photochemical Green Synthesis of Water-Soluble Ag, Au and Au@Ag Core-Shell Nanoparticles, Chem. Eur. J. 2019, 25, 14638-14643.
- J. Garcia-Amoròs, S. Swaminathan, S. Sortino and F. M. Raymo, Plasmonic activation of a fluorescent carbazole-oxazine switch, Chem. Eur. J. 2014, 20, 10276-10284.
- S. Giuffrida, G. Ventimiglia and S. Sortino, Straightforward Green Synthesis of “naked” aqueous silver nanoparticles, Chem. Commun., 2009, 27, 4055-4057.
- E. B. Caruso, S. Petralia, S. Conoci, S. Giuffrida and S. Sortino, Photodelivery of Nitric Oxide from Water-Soluble Platinum Nanoparticles, J. Am. Chem. Soc. 2007 (129) 480-481.
